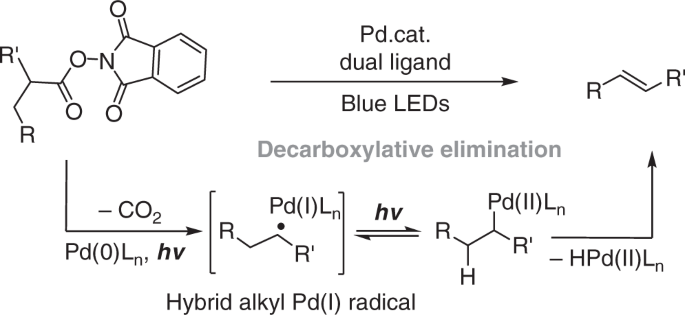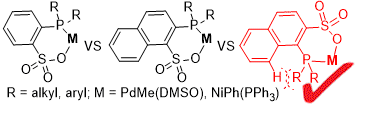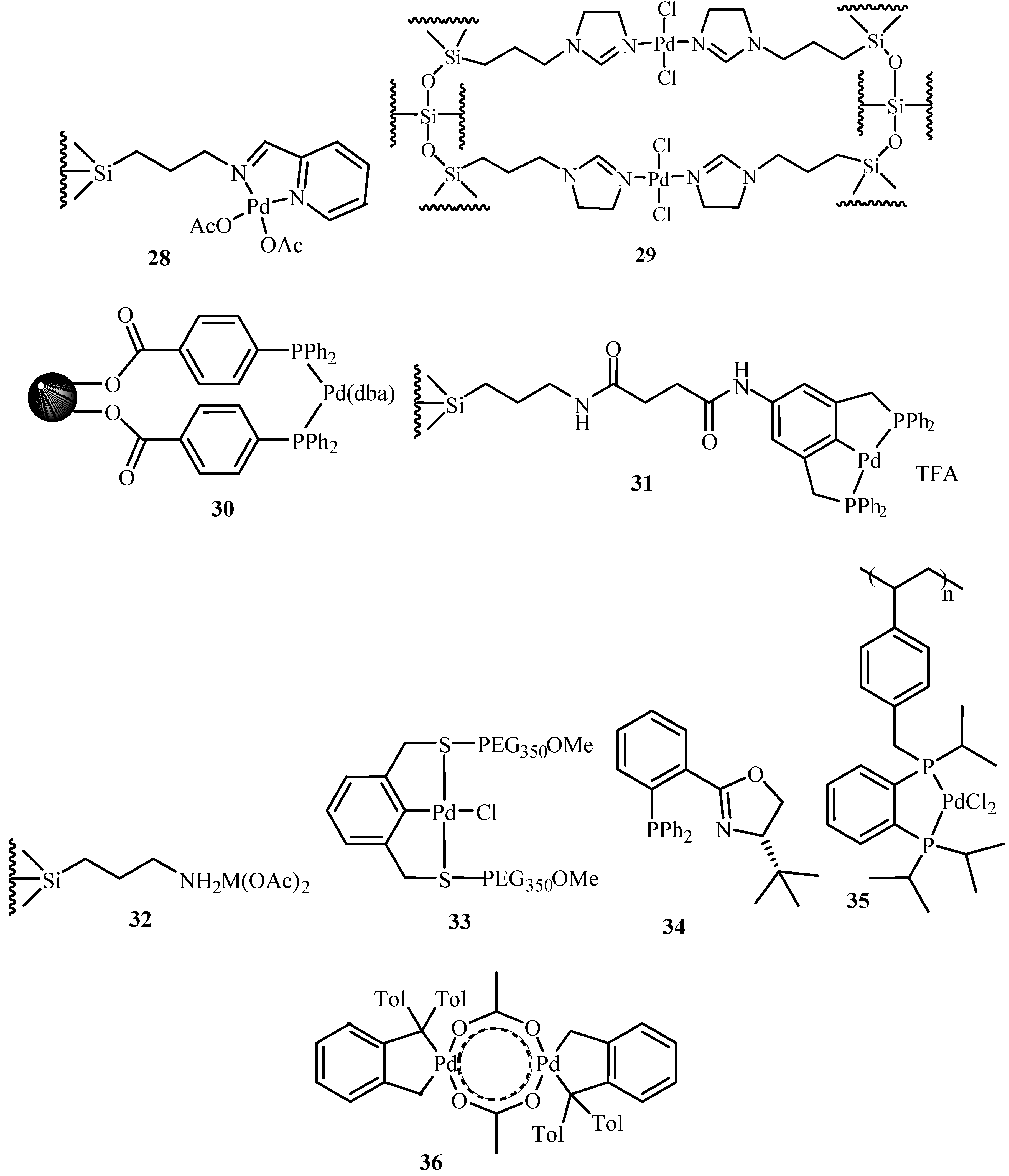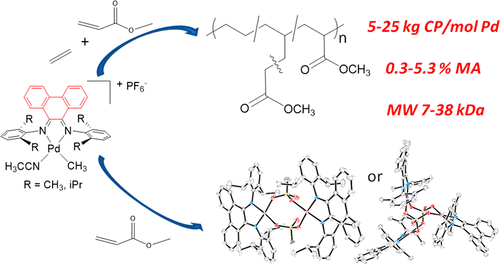
Palladium-Catalyzed Ethylene/Methyl Acrylate Copolymerization: Moving from the Acenaphthene to the Phenanthrene Skeleton of α-Diimine Ligands,Organometallics - X-MOL

Selective Ethylene Dimerization by Palladium(II) Complexes Bearing a Phosphinoferrocene Sulfonate Ligand,Organometallics - X-MOL
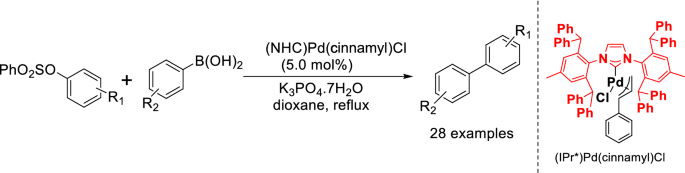
Pd(NHC)(cinnamyl)Cl-catalyzed Suzuki cross-coupling reaction of aryl sulfonates with arylboronic acids | SpringerLink

Sodium 2-(2-pyridin-3-ylethylamino)sulfonate: an efficient ligand and base for palladium-catalyzed Heck reaction in aqueous media - ScienceDirect

Synthesis and Catalytic Properties of Cyclopalladated Complexes Bearing a Phosphane‐Sulfonate Ligand - Eur. J. Inorg. Chem. - X-MOL

Recent Advances in the Palladium Catalyzed Suzuki–Miyaura Cross-Coupling Reaction in Water | SpringerLink
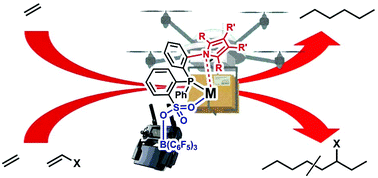
Ligand–metal secondary interactions in phosphine–sulfonate palladium and nickel catalyzed ethylene (co)polymerization - Polymer Chemistry (RSC Publishing)
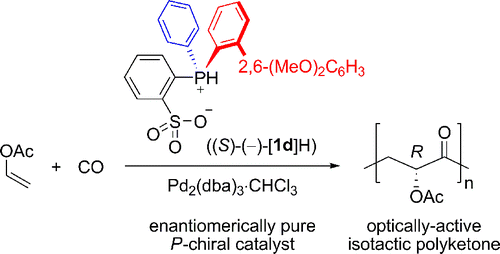
P-chiral phosphine-sulfonate/palladium-catalyzed asymmetric copolymerization of vinyl acetate with carbon monoxide. - J. Am. Chem. Soc. - X-MOL

Figure 3 from Ortho-phosphinobenzenesulfonate: a superb ligand for palladium-catalyzed coordination-insertion copolymerization of polar vinyl monomers. | Semantic Scholar

Synthesis and Catalytic Properties of Cyclopalladated Complexes Bearing a Phosphane‐Sulfonate Ligand - Dong - 2019 - European Journal of Inorganic Chemistry - Wiley Online Library
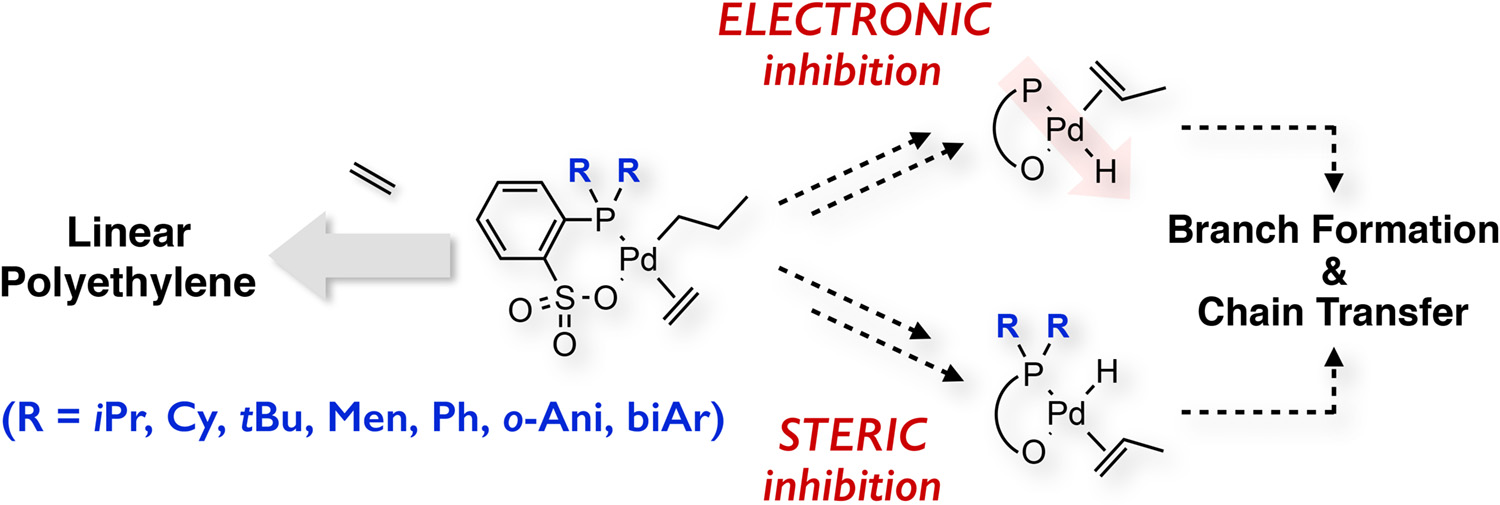
Elucidating the Key Role of Phosphine–Sulfonate Ligands in Palladium-Catalyzed Ethylene Polymerization: Effect of Ligand Structure on the Molecular Weight and Linearity of Polyethylene - Ito Research Group
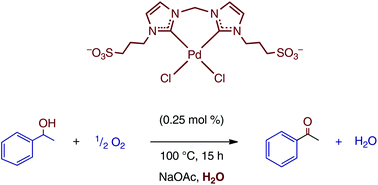
Synthesis of water-soluble palladium(ii) complexes with N-heterocyclic carbene chelate ligands and their use in the aerobic oxidation of 1-phenylethanol - Dalton Transactions (RSC Publishing)
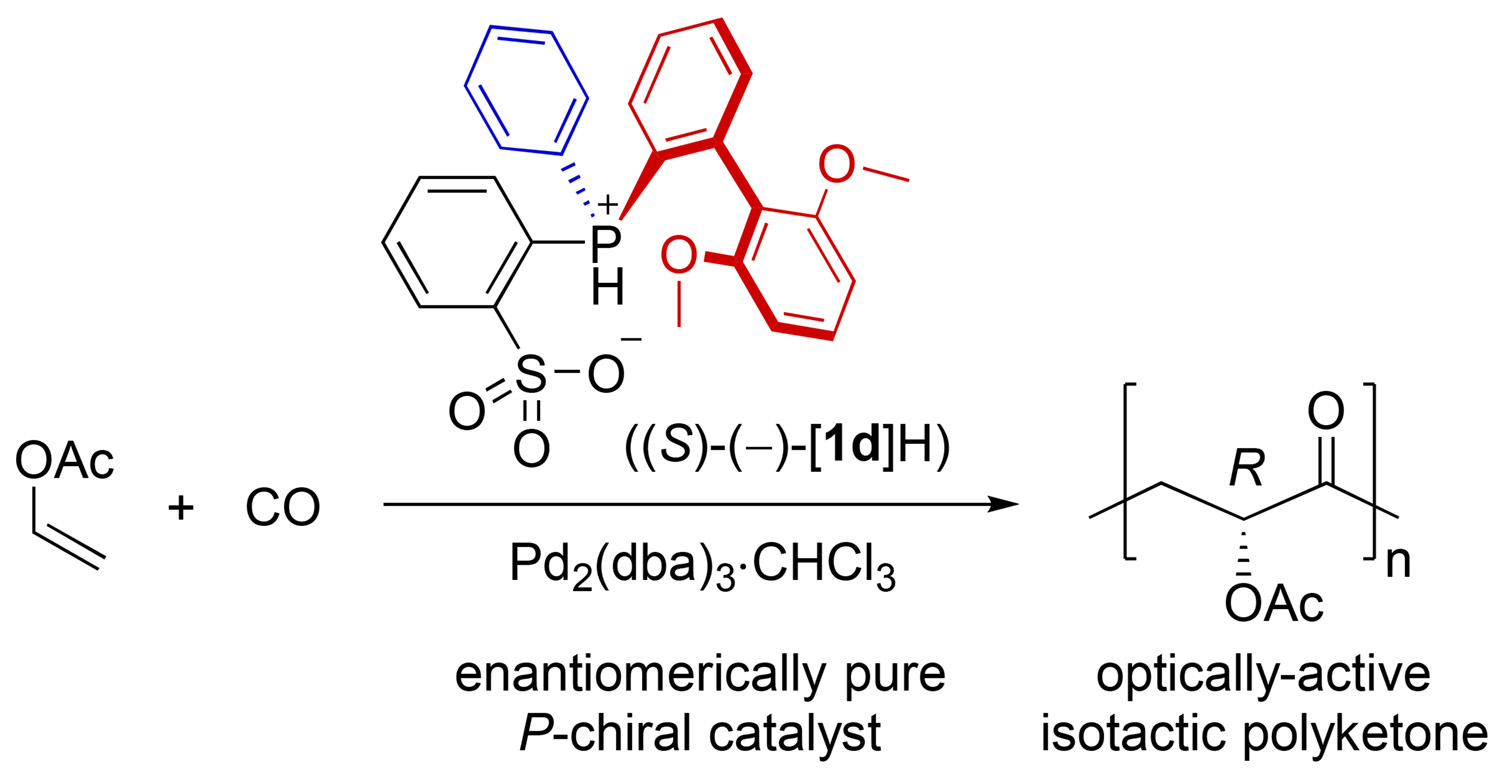
P-Chiral Phosphine–Sulfonate/Palladium-Catalyzed Asymmetric Copolymerization of Vinyl Acetate with Carbon Monoxide - Ito Research Group

Palladium Catalysts with Sulfonate-Functionalized-NHC Ligands for Suzuki−Miyaura Cross-Coupling Reactions in Water – topic of research paper in Chemical sciences. Download scholarly article PDF and read for free on CyberLeninka open science hub.
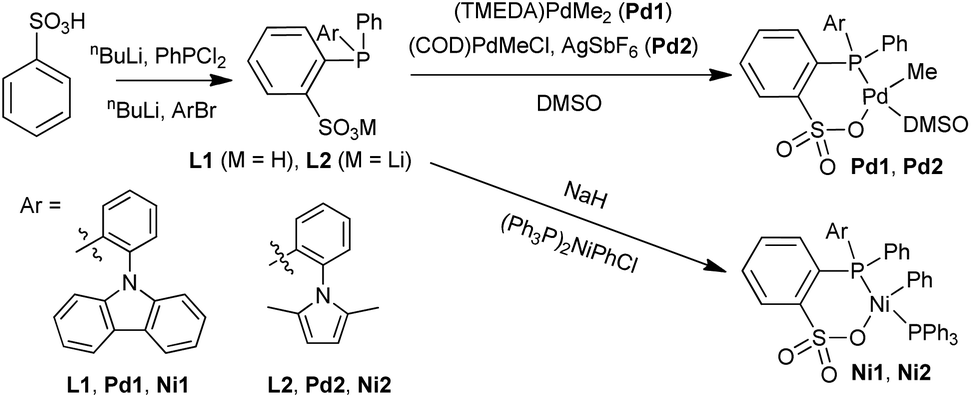
Ligand–metal secondary interactions in phosphine–sulfonate palladium and nickel catalyzed ethylene (co)polymerization - Polymer Chemistry (RSC Publishing)
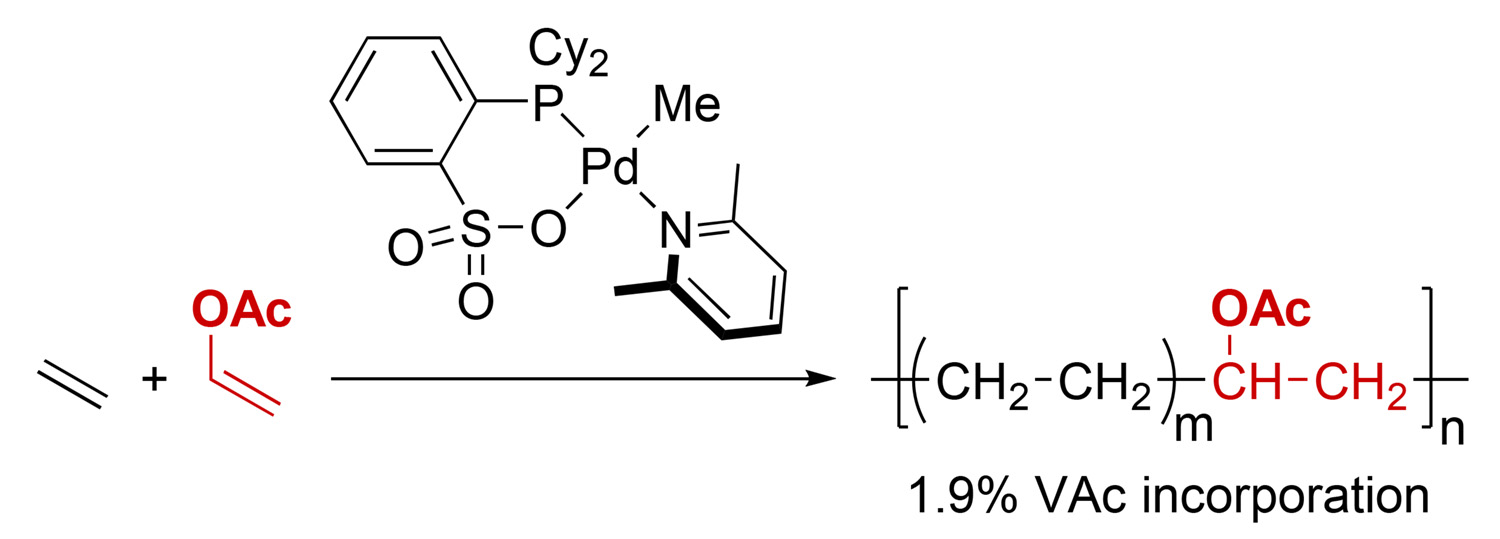
Copolymerization of Vinyl Acetate with Ethylene by Palladium/Alkylphosphine– Sulfonate Catalysts - Ito Research Group