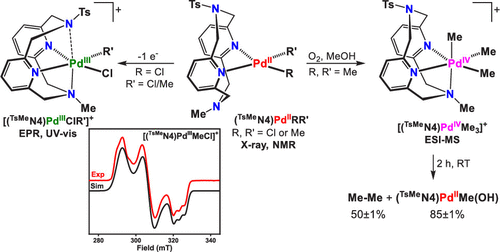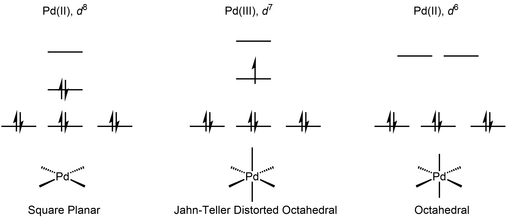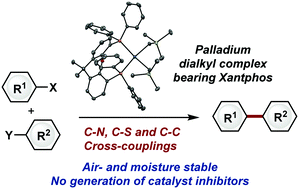
Air- and moisture-stable Xantphos-ligated palladium dialkyl complex as a precatalyst for cross-coupling reactions - Chemical Communications (RSC Publishing)

Functionalized nitrogen ligands for palladium catalyzed cross-coupling reactions (part I) - ScienceDirect

Norbornene polymerization and copolymerization with 1-alkenes by neutral palladium complexes bearing aryloxide imidazolin-2-imine ligand† - Polym. Chem. - X-MOL
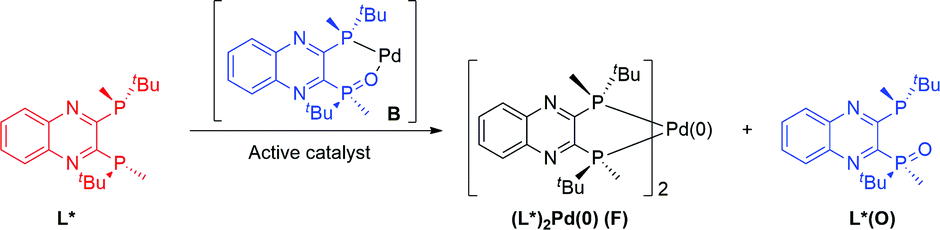
A rational pre-catalyst design for bis-phosphine mono-oxide palladium catalyzed reactions - Chemical Science (RSC Publishing) DOI:10.1039/C6SC05472B

Irradiation-induced palladium-catalyzed decarboxylative desaturation enabled by a dual ligand system | Nature Communications
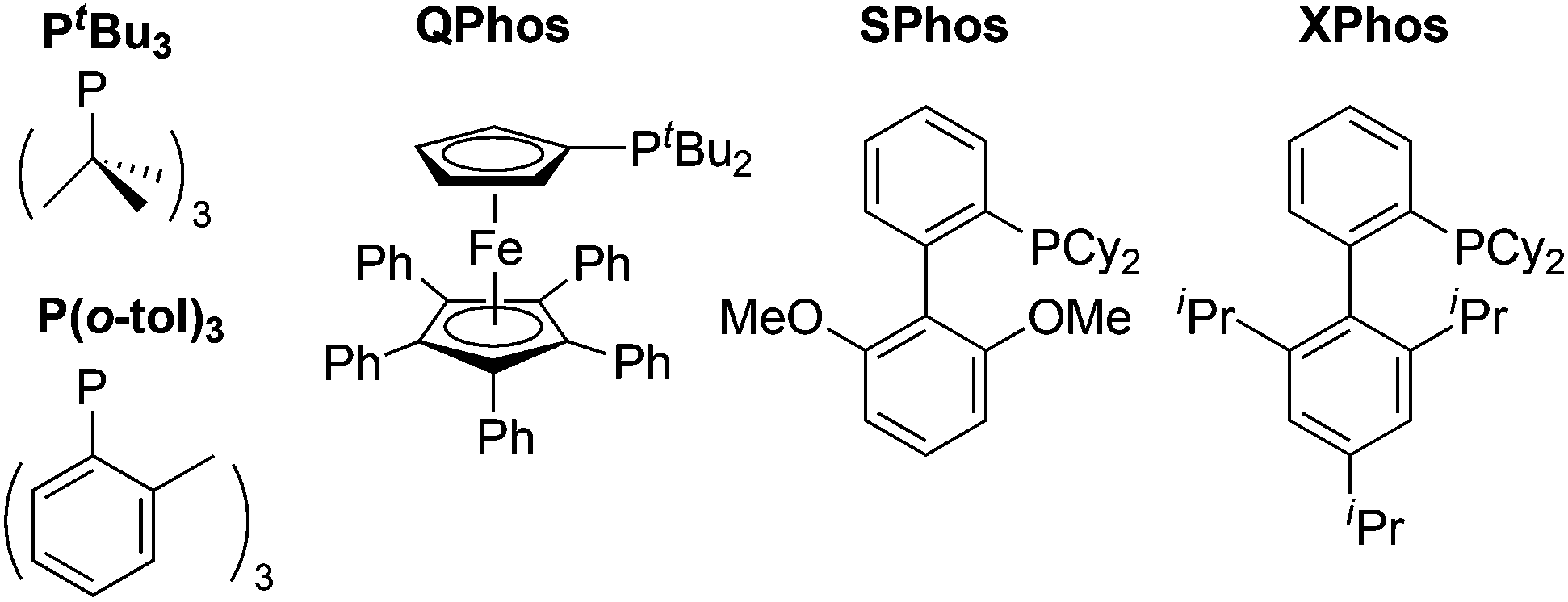
Computed ligand effects on the oxidative addition of phenyl halides to phosphine supported palladium(0) catalysts - Dalton Transactions (RSC Publishing) DOI:10.1039/C4DT01758G
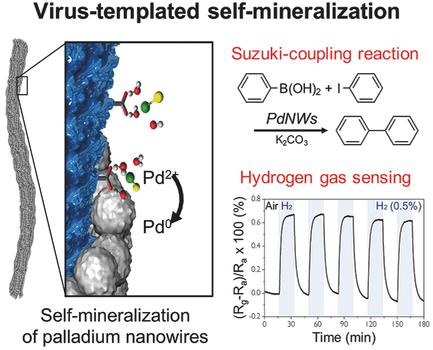
Virus‐Templated Self‐Mineralization of Ligand‐Free Colloidal Palladium Nanostructures for High Surface Activity and Stability - Adv. Funct. Mater. - X-MOL
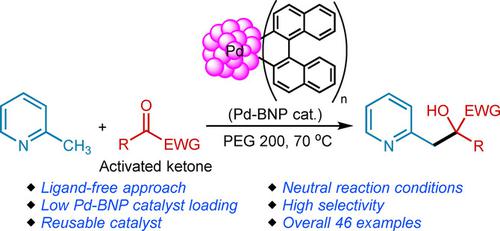
Ligand‐Free and Reusable Palladium Nanoparticles‐Catalyzed Alkylation of 2‐Alkylazaarenes with Activated Ketones under Neutral Conditions - Adv. Synth. Catal. - X-MOL
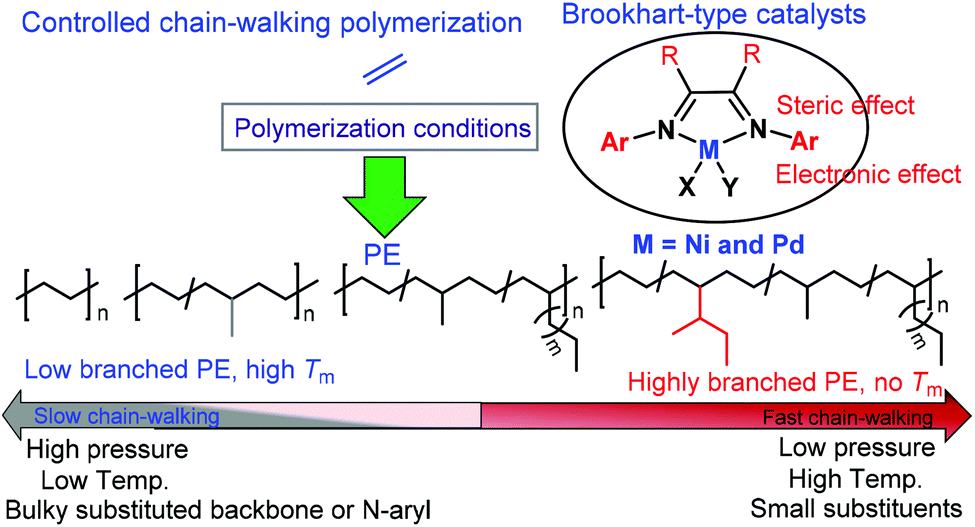
A continuing legend: the Brookhart-type α-diimine nickel and palladium catalysts - Polymer Chemistry (RSC Publishing)

Superstable Palladium(0) Complex as an Air‐ and Thermostable Catalyst for Suzuki Coupling Reactions - Jakab - 2015 - European Journal of Organic Chemistry - Wiley Online Library
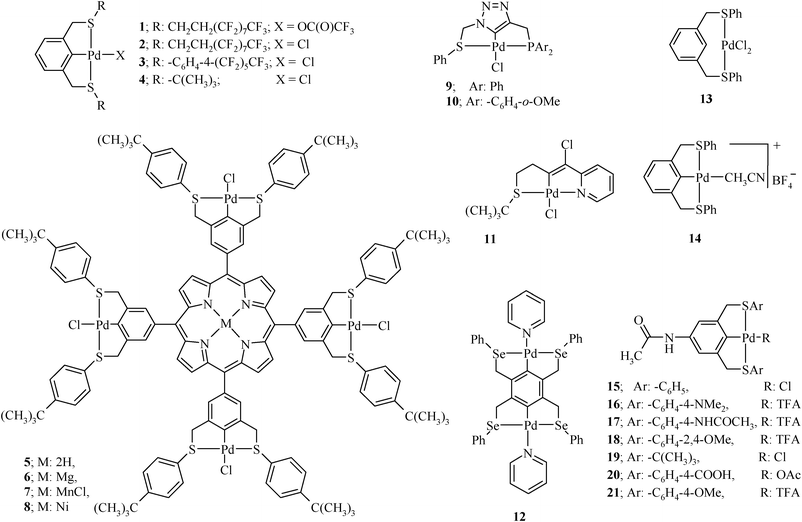
Organochalcogen ligands and their palladium( ii ) complexes: Synthesis to catalytic activity for Heck coupling - RSC Advances (RSC Publishing) DOI:10.1039/C2RA20508D
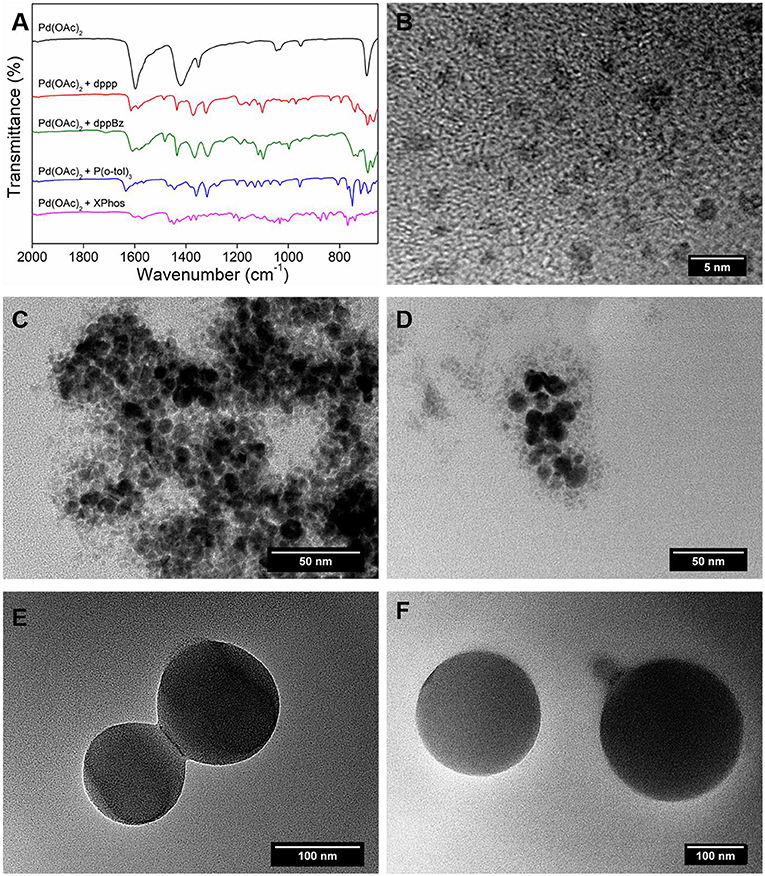
Frontiers | Ligand and Solvent Selection for Enhanced Separation of Palladium Catalysts by Organic Solvent Nanofiltration | Chemistry

PDF) Palladium(II) halide complexes. I. Stabilities and spectra of palladium(II) chloro and bromo aqua complexes

High in vitro anticancer activity of a dinuclear palladium(II) complex with a 2‑phenylpyridine ligand - ScienceDirect
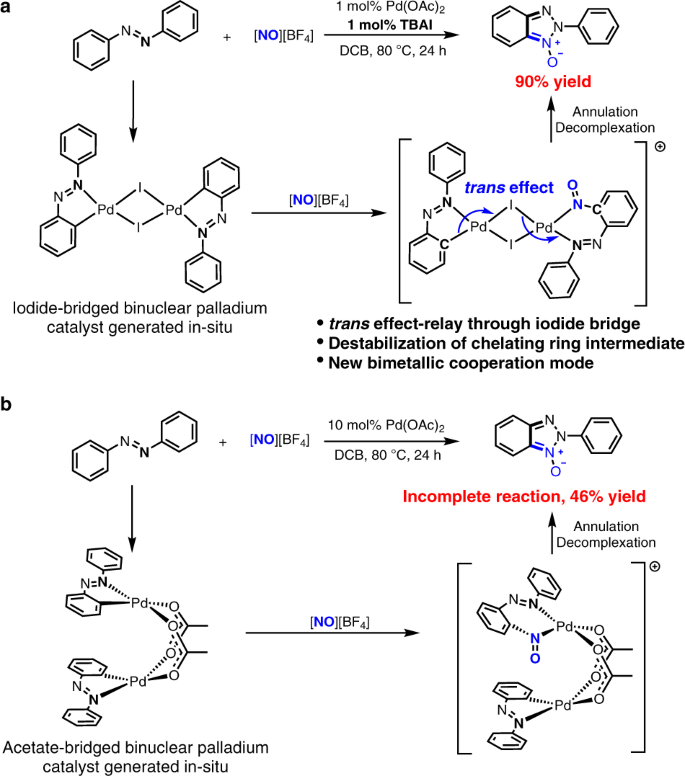
Iodide-enhanced palladium catalysis via formation of iodide-bridged binuclear palladium complex | Communications Chemistry
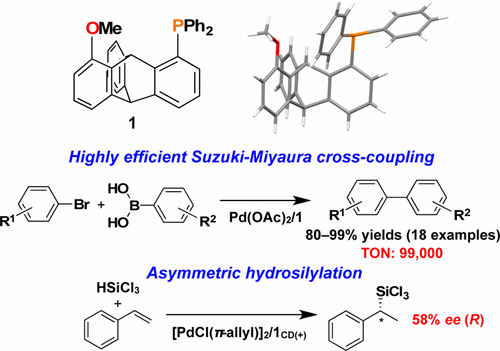
Synthesis and Catalytic Applications of a Triptycene-Based Monophosphine Ligand for Palladium-Mediated Organic Transformations. - ACS Omega - X-MOL