
Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

Thiol Treatment Creates Selective Palladium Catalysts for Semihydrogenation of Internal Alkynes - ScienceDirect

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

Palladium-catalyzed carbon-sulfur or carbon-phosphorus bond metathesis by reversible arylation | Science
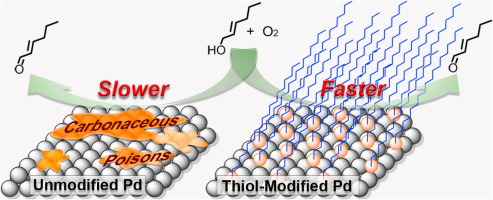
Application of thiolate self-assembled monolayers in selective alcohol oxidation for suppression of Pd catalyst deactivation - J. Catal. - X-MOL

Thiol Treatment Creates Selective Palladium Catalysts for Semihydrogenation of Internal Alkynes - ScienceDirect
Tuning the Catalytic Activity and Selectivity of Pd Nanoparticles Using Ligand-Modified Supports and Surfaces
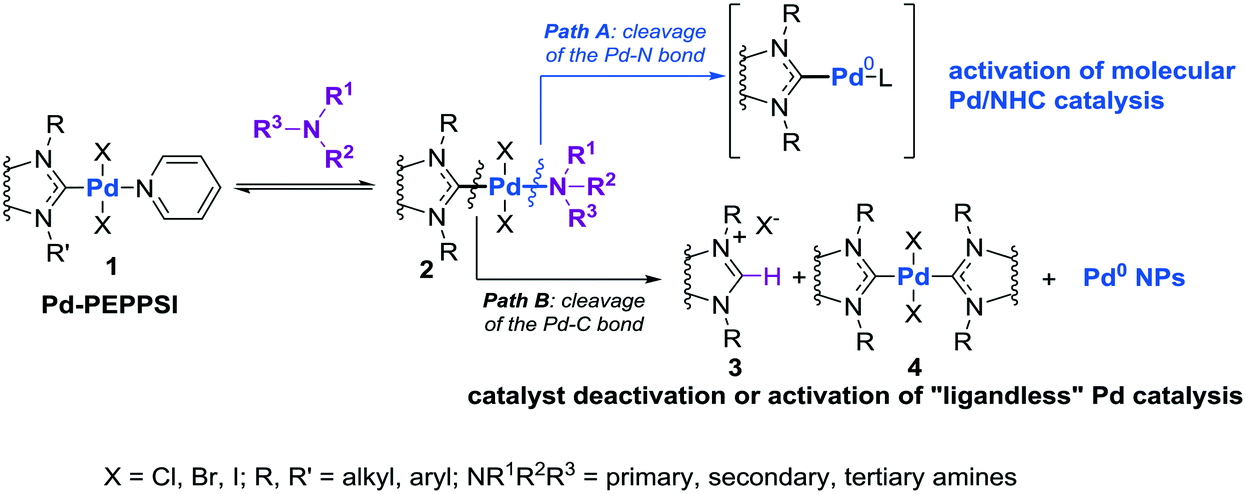
Preventing Pd–NHC bond cleavage and switching from nano-scale to molecular catalytic systems: amines and temperature as catalyst activators - Catalysis Science & Technology (RSC Publishing)

Optimising surface d charge of AuPd nanoalloy catalysts for enhanced catalytic activity | Nature Communications
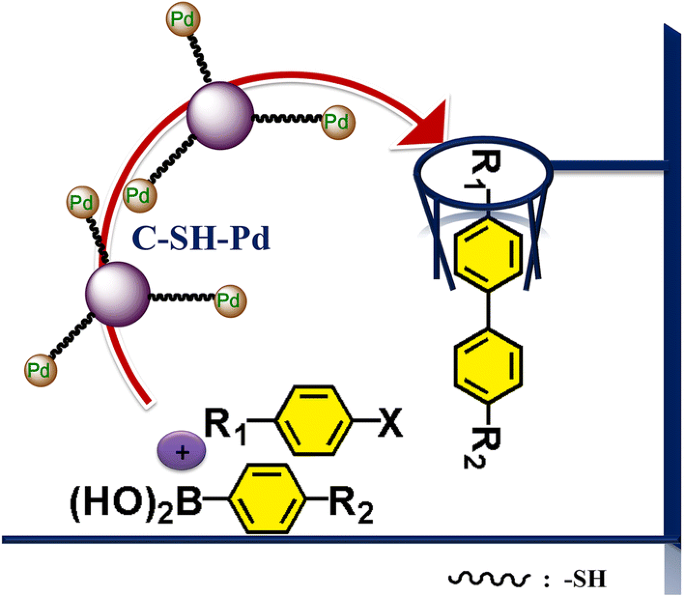
Palladium Nanoparticles Anchored on Thiol Functionalized Xylose Hydrochar Microspheres: An Efficient Heterogeneous Catalyst for Suzuki Cross-Coupling Reactions | SpringerLink
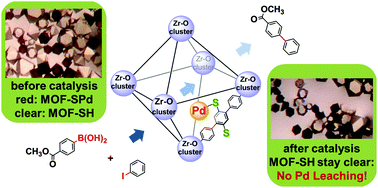
Tackling poison and leach: catalysis by dangling thiol–palladium functions within a porous metal–organic solid - Chemical Communications (RSC Publishing)
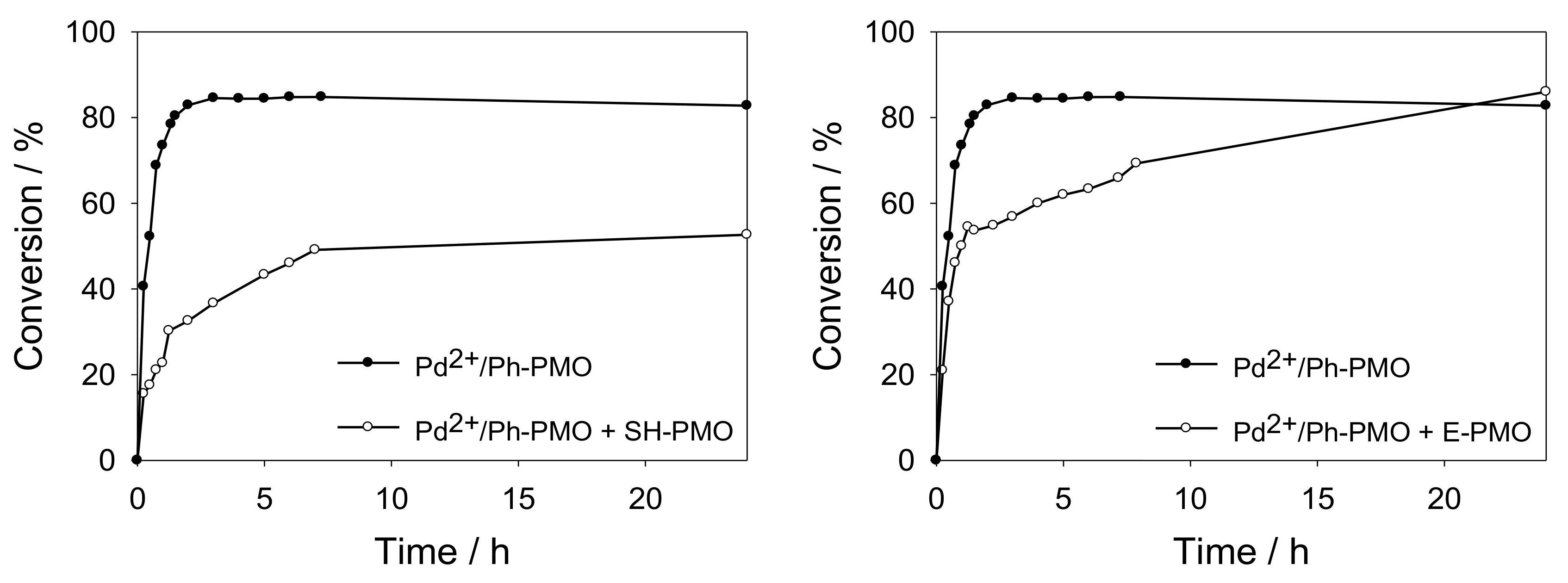
Materials | Free Full-Text | Thiol-Functionalized Ethylene Periodic Mesoporous Organosilica as an Efficient Scavenger for Palladium: Confirming the Homogeneous Character of the Suzuki Reaction | HTML

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds
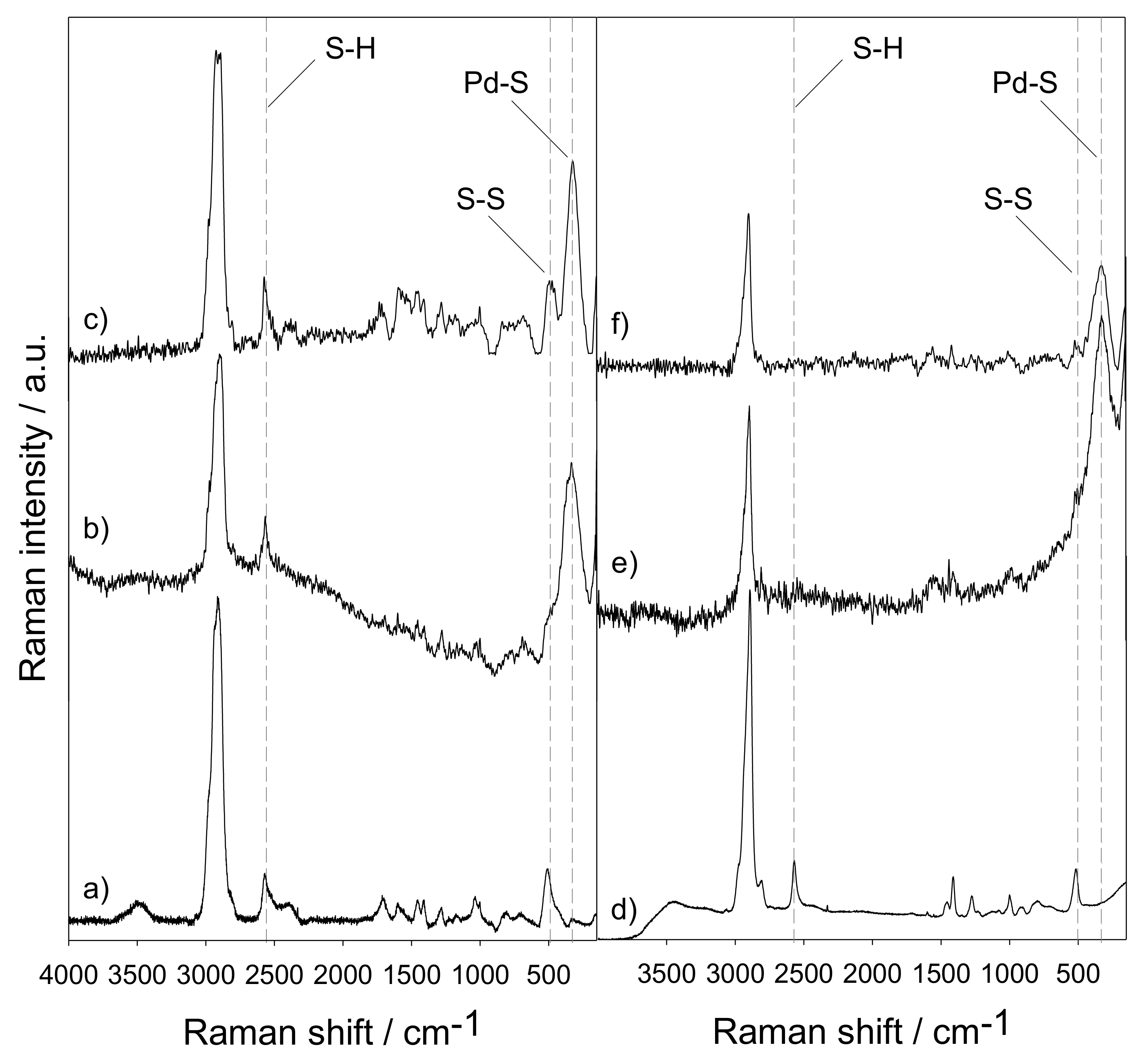
Materials | Free Full-Text | Thiol-Functionalized Ethylene Periodic Mesoporous Organosilica as an Efficient Scavenger for Palladium: Confirming the Homogeneous Character of the Suzuki Reaction | HTML

Palladium‐Catalyzed C–S Bond Formation of Stable Enamines with Arene/Alkanethiols: Highly Regioselective Synthesis of β‐Amino Sulfides - Jiang - 2016 - European Journal of Organic Chemistry - Wiley Online Library
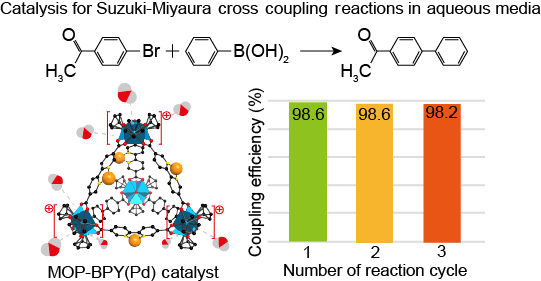
Single-atom Pd catalyst anchored on Zr-based metal-organic polyhedra for Suzuki-Miyaura cross coupling reactions in aqueous media - Nano Res. - X-MOL
Selective ensembles in supported palladium sulfide nanoparticles for alkyne semi-hydrogenation | Nature Communications

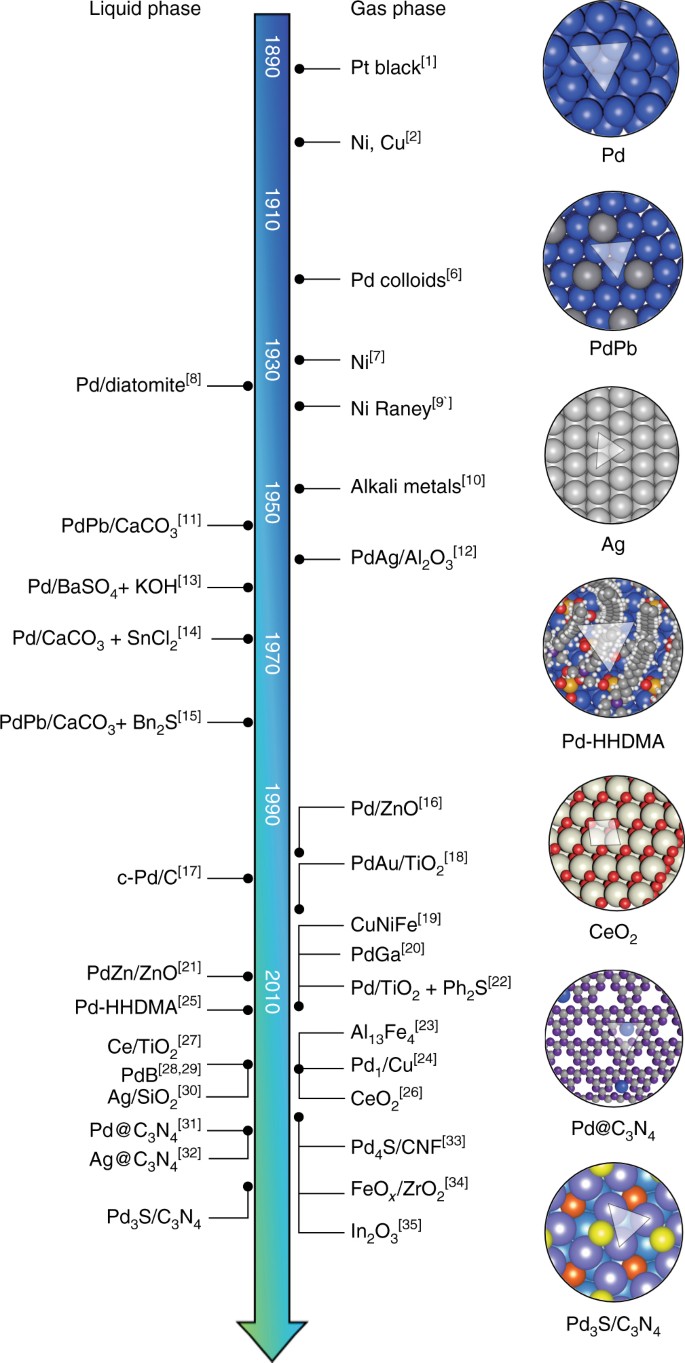
The structure of magnetically-supported palladium catalysts used in... | Download Scientific Diagram


