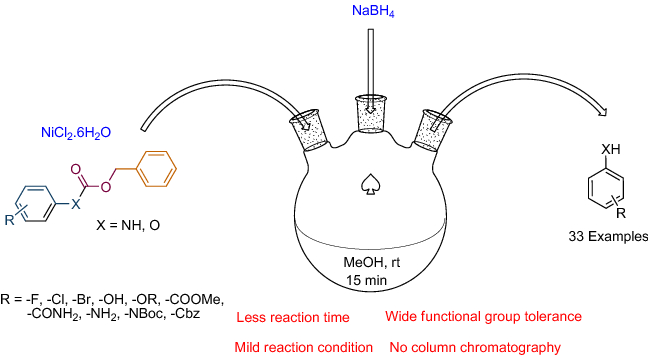
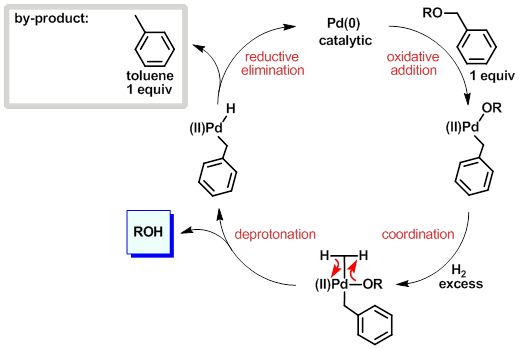
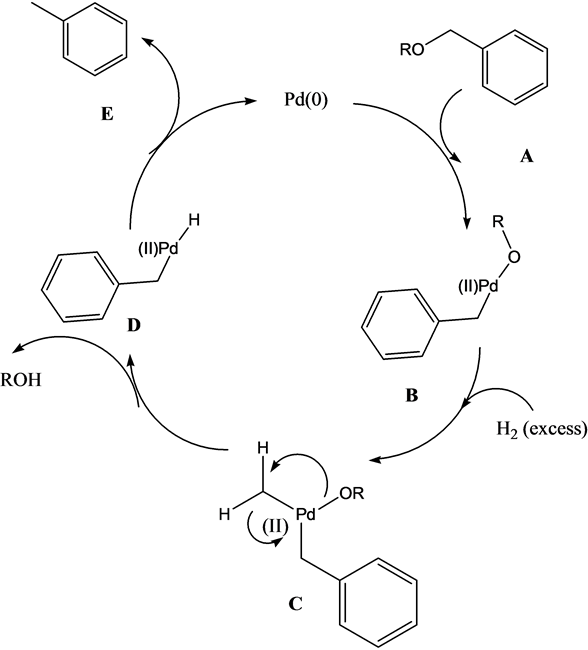A Mechanistic Study of Direct Activation of Allylic Alcohols in Palladium Catalyzed Amination Reactions

Mild Palladium‐Catalyzed Cyanation of Unprotected 2‐Iodoglycals in Aqueous Media as Versatile Tool to Access Diverse C2‐Glycoanalogues - Malinowski - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library

Facile and selective cleavage of allyl ethers, amines and esters using polymethylhydrosiloxane–ZnCl2/Pd(PPh3)4 - ScienceDirect

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds
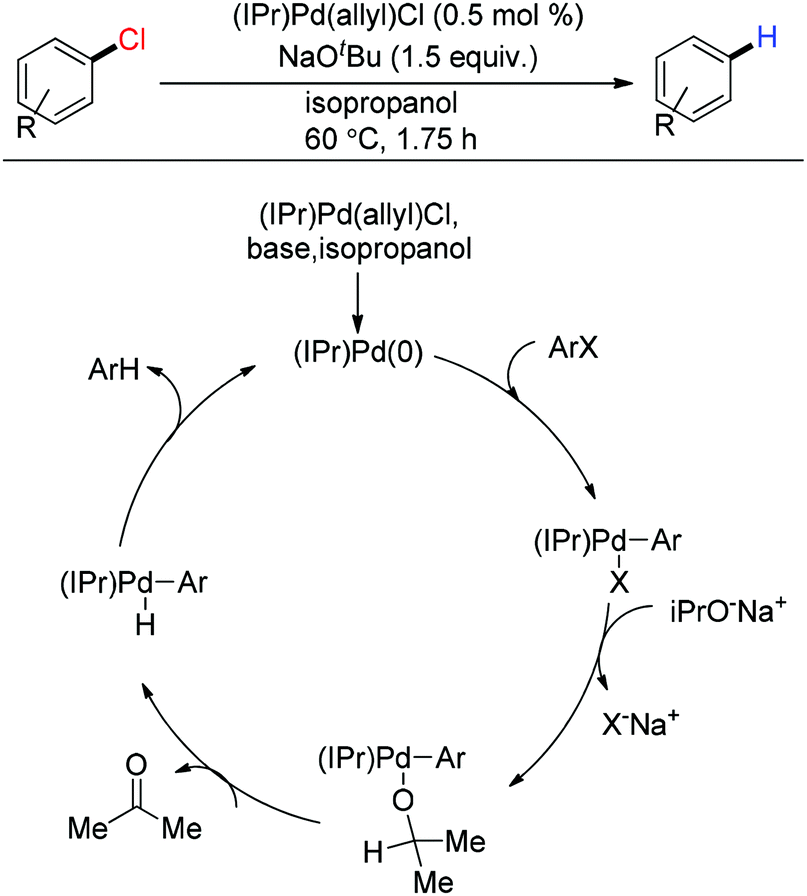
Metal catalyzed defunctionalization reactions - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C5OB01949D

Microwave‐assisted cleavage of Alloc and Allyl Ester protecting groups in solid phase peptide synthesis - Wilson - 2016 - Journal of Peptide Science - Wiley Online Library
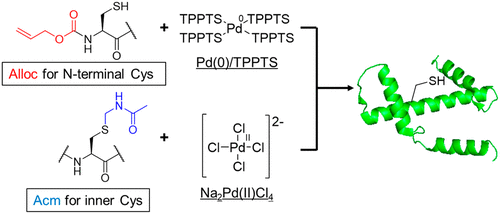
Chemical Synthesis of Cys-Containing Protein via Chemoselective Deprotection with Different Palladium Complexes.,Organic Letters - X-MOL
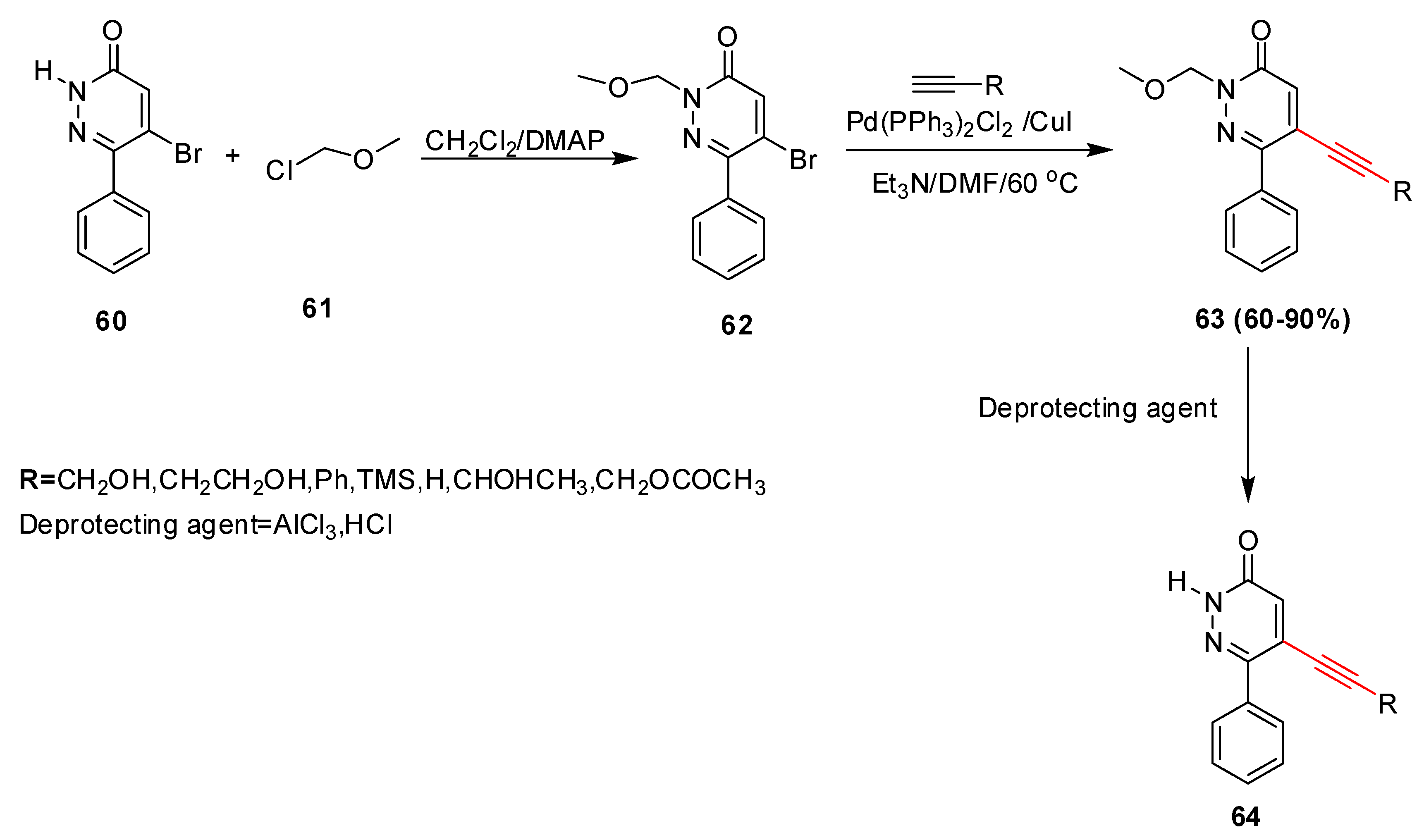
Catalysts | Free Full-Text | Palladium and Copper Catalyzed Sonogashira cross Coupling an Excellent Methodology for C-C Bond Formation over 17 Years: A Review | HTML

Palladium-triggered deprotection chemistry for protein activation in living cells | Nature Chemistry

Palladium-catalyzed reaction of tributyltin hydride. Selective and very mild deprotection of allyl and allyloxycarbonyl derivatives of amino-acids

Intracellular Deprotection Reactions Mediated by Palladium Complexes Equipped with Designed Phosphine Ligands. - Abstract - Europe PMC

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

Intracellular Deprotection Reactions Mediated by Palladium Complexes Equipped with Designed Phosphine Ligands. - Abstract - Europe PMC

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

Microwave‐assisted cleavage of Alloc and Allyl Ester protecting groups in solid phase peptide synthesis - Wilson - 2016 - Journal of Peptide Science - Wiley Online Library