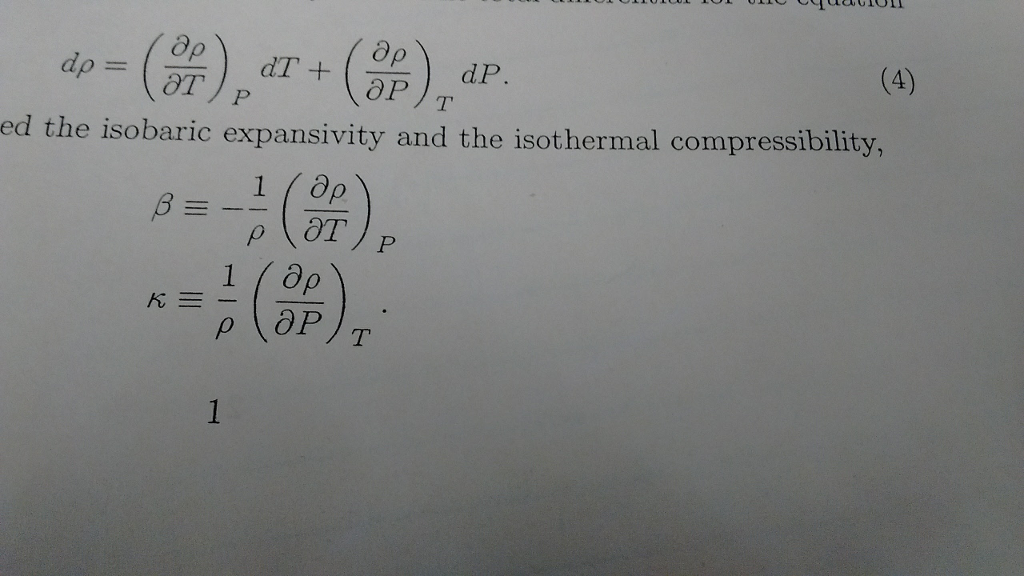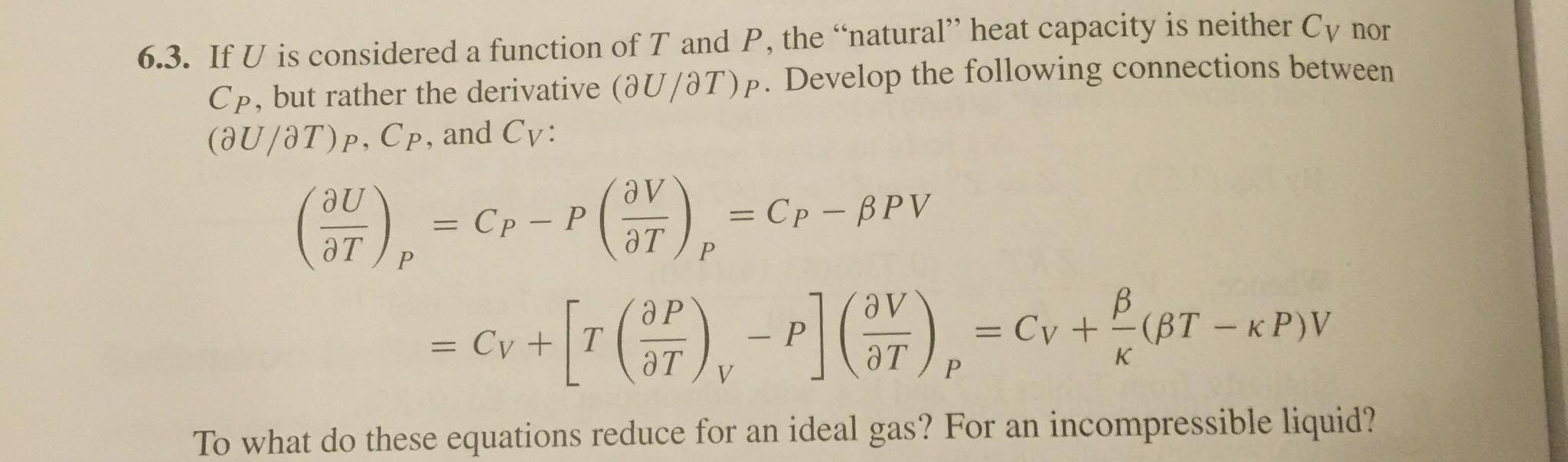Department of Mechanical Engineering ME 322 – Mechanical Engineering Thermodynamics Lecture 5 Thermodynamic Properties. - ppt download
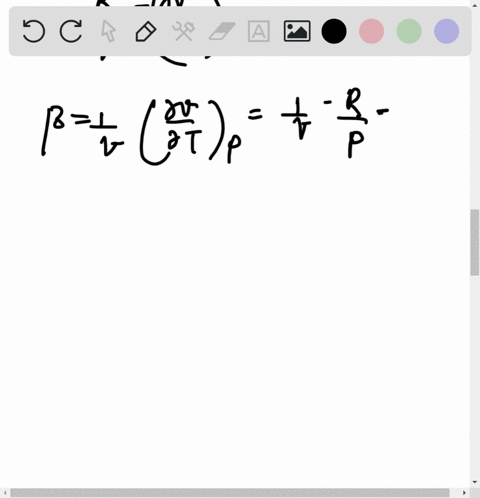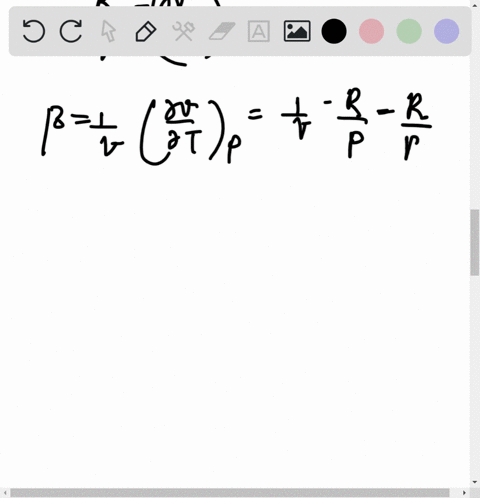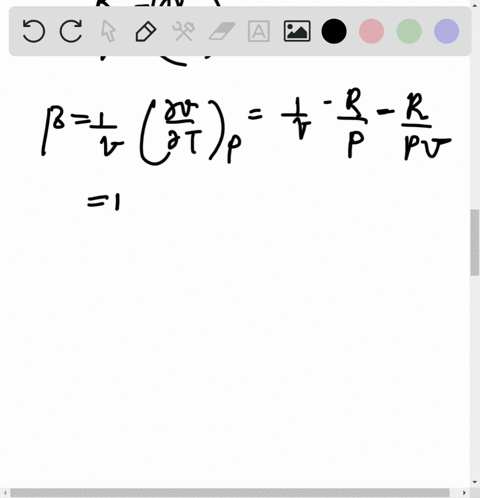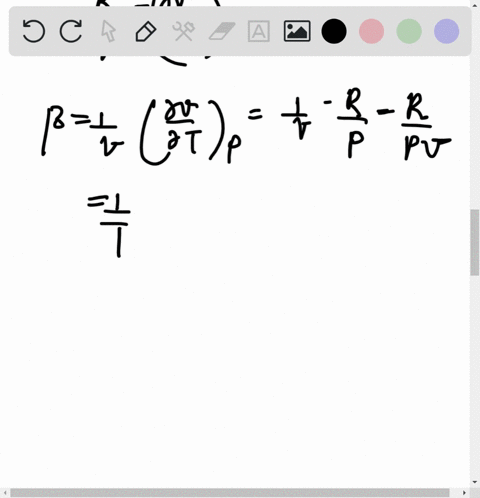
SOLVED:Develop expressions for the volume expansivity \beta and the isothermal compressibility \kappa for (a) an ideal gas. (b) a gas whose equation of state is p(v-b)=R T. (c) a gas obeying the

The compressibility kappa of a substance is defined as the fractional change in volume of that substance for a given change in pressure : kappa = - 1V dVdP (a) Explain why
![Calculate the isothermal compressibility 'alpha' for an ideal gas [alpha = - (1)/(V) ((deltaV)/(deltaV))(T.n)]at P=1 bar and cubic expansion coefficient beta [beta = - (1)/(V) ((deltaP)/(deltaT))(P.n)]"for an ideal " gas at T = Calculate the isothermal compressibility 'alpha' for an ideal gas [alpha = - (1)/(V) ((deltaV)/(deltaV))(T.n)]at P=1 bar and cubic expansion coefficient beta [beta = - (1)/(V) ((deltaP)/(deltaT))(P.n)]"for an ideal " gas at T =](https://d10lpgp6xz60nq.cloudfront.net/question-thumbnail/en_201230540.png)
Calculate the isothermal compressibility 'alpha' for an ideal gas [alpha = - (1)/(V) ((deltaV)/(deltaV))(T.n)]at P=1 bar and cubic expansion coefficient beta [beta = - (1)/(V) ((deltaP)/(deltaT))(P.n)]"for an ideal " gas at T =

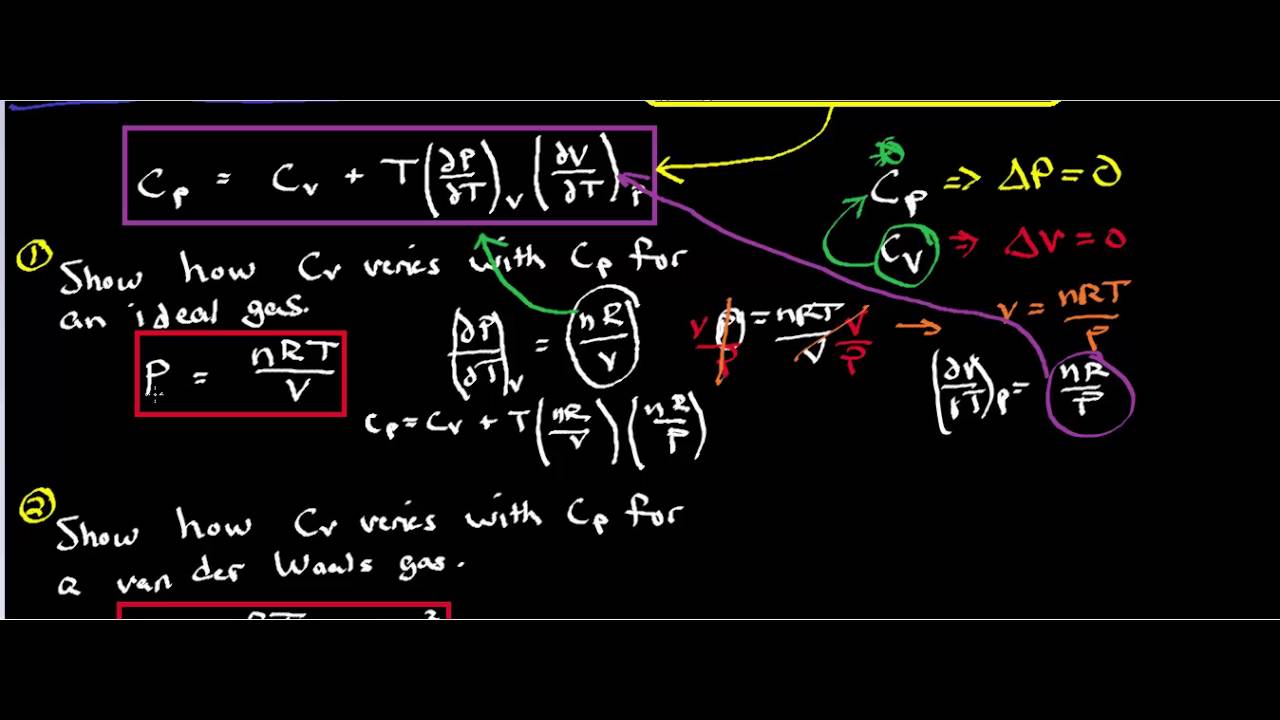
Cp-Cv for real gas, in terms of alpha & beta, change in internal energy with respect to volume - YouTube
![Calculate the isothermal compressibility 'alpha' for an ideal gas [alpha = - (1)/(V) ((deltaV)/(deltaV))(T.n)]at P=1 bar and cubic expansion coefficient beta [beta = - (1)/(V) ((deltaP)/(deltaT))(P.n)]"for an ideal " gas at T = Calculate the isothermal compressibility 'alpha' for an ideal gas [alpha = - (1)/(V) ((deltaV)/(deltaV))(T.n)]at P=1 bar and cubic expansion coefficient beta [beta = - (1)/(V) ((deltaP)/(deltaT))(P.n)]"for an ideal " gas at T =](https://doubtnut-static.s.llnwi.net/static/web-thumb/201230540_web.png)